The Digital Liver Lab (DiLiLab) of the Institute of Structural Mechanics and Dynamics in Aerospace Engineering deals with the multiscale and multiphase description of the liver based on the Theory of Porous Media (TPM). The key aspect of knowledge-driven modeling is the continuum-biomechanical description of the lobular level coupled with a systems biology approach at the cellular level while taking into account the blood pressure at the organ level. In addition, data-driven surrogate modeling applications are investigated.
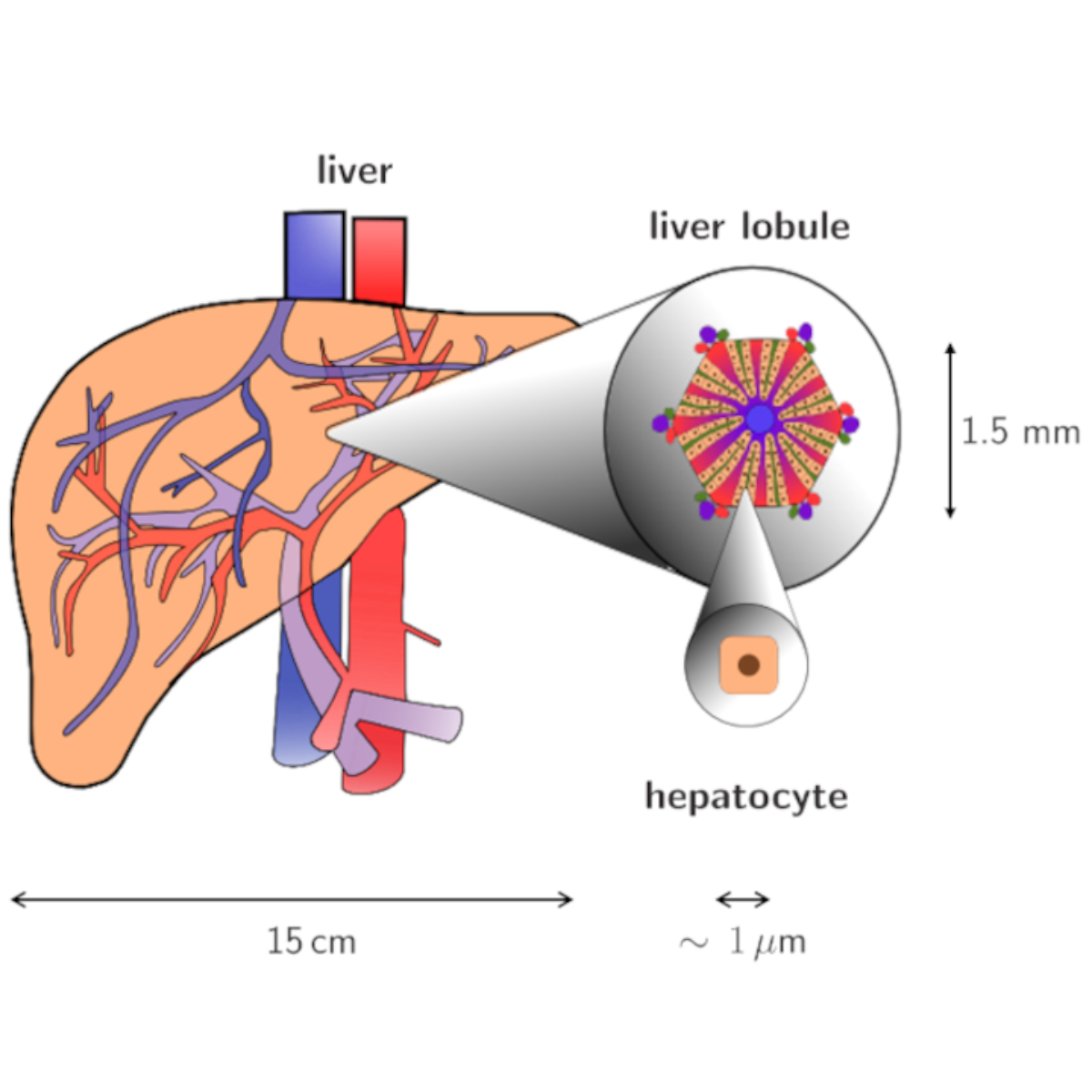
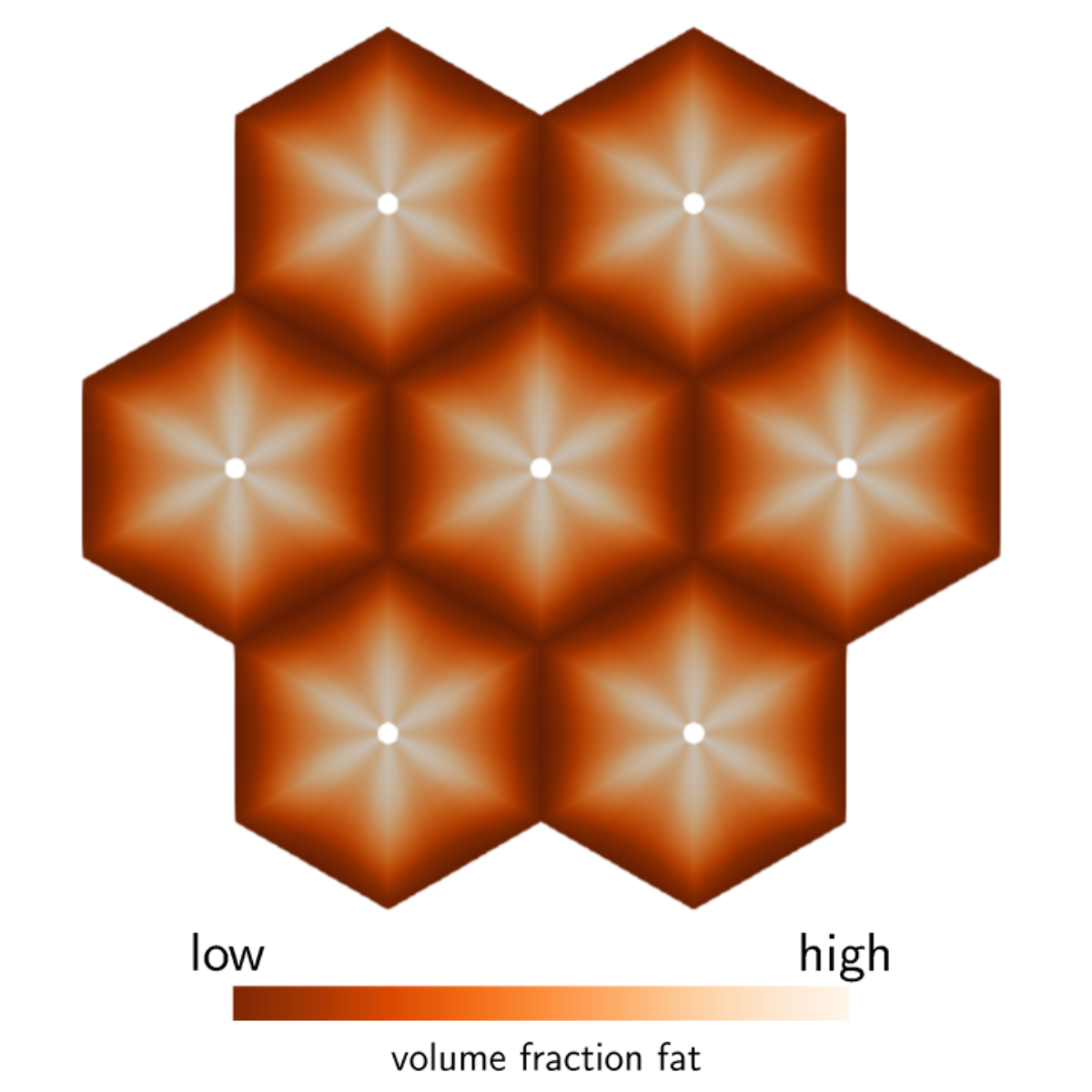
The liver is the central metabolic organ in the human body. The main tasks of the liver are metabolic homeostasis, e.g., lipid and carbohydrate metabolism, detoxification of drugs and toxins and storage as well as depletion of high-emergy substances. The functionality of the liver is mainly influenced by the perfusion of the smallest repetitive sub-elements, the so-called lobules. A dysfunctional perfusion is mainly caused by diseases of the liver, such as steatosis, cirrhosis or tumor growth as well as surgical interventions like (extended) liver resection or liver transplantation.
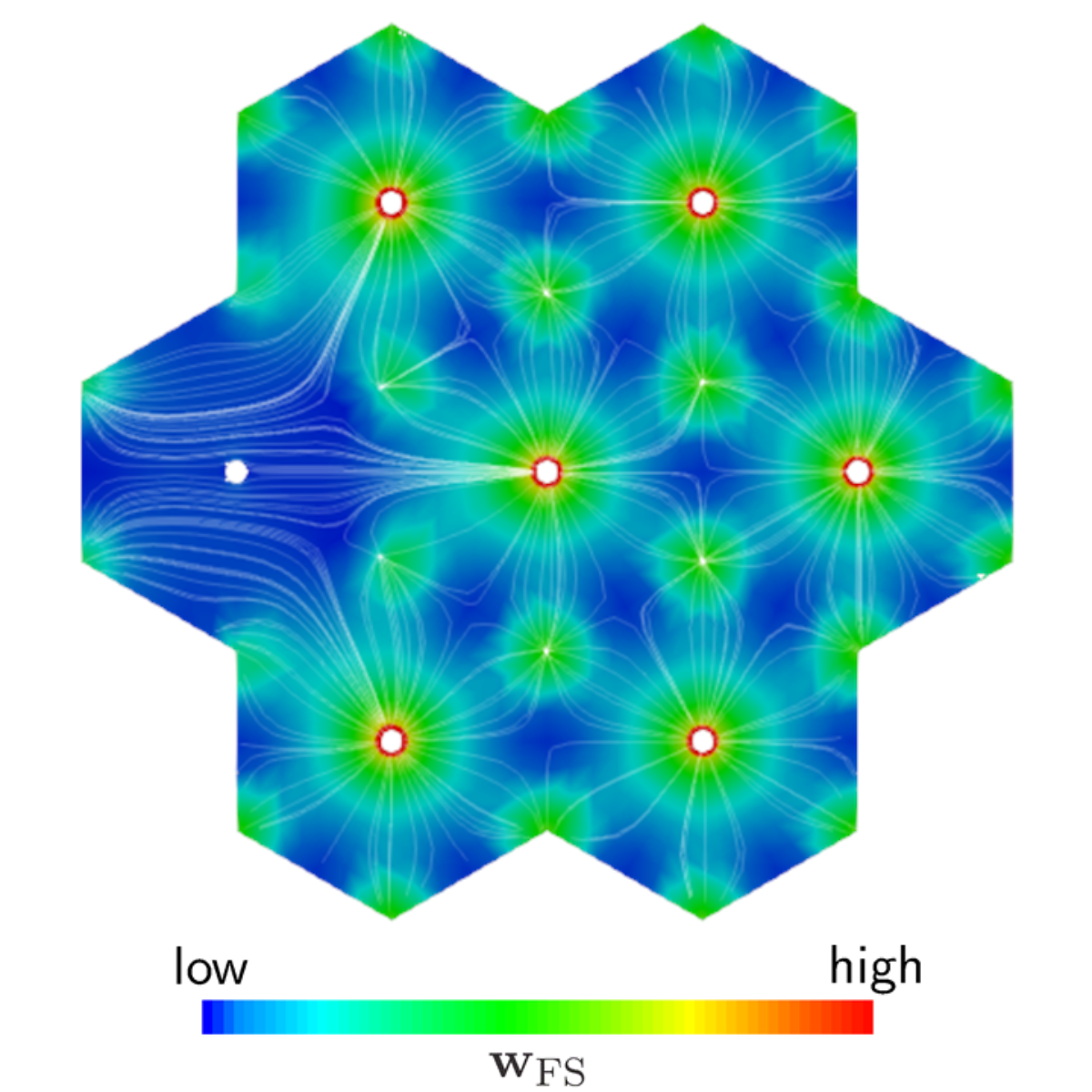
To simulate the processes in the liver, we use a multicomponent, poro-elastic, multiphasic and multiscale function-perfusion approach. On the organ scale, the total perfusion as well as the blood pressure is calculated. This affects the blood perfusion through the liver lobules on the lobule scale, where we have an anisotropic blood flow along the sinusoid, the liver cells. On the lobule scale, the complex biological structure of the liver lobules is described by the Theory of Porous Media. The metabolism processes take place on the cell scale, where we use ordinary differential equations to describe the extensive procedures.
Using this multiphasic, continuum-mechanical formulation of the liver, taking the macro scale (organ level), meso scale (liver lobule) and micro scale (metabolism in the hepatocytes) into account, various disorders and therapeutic procedures in addition to the general function can be described. This numerical model allows a non-invasive, patient-specific analysis of the liver and can help to improve therapy as well as actual surgical procedures.
All completed and ongoing projects are listed below.
Cooperation Partners
- Matthias König, Humboldt Universität Berlin
- Uta Dahmen, Uniklinikum Jena
- Hans-Michael Tautenhahn, Uniklinikum Jena
- Nicole Radde, Universität Stuttgart
- Lars Ole Schwen, MEVIS Bremen
- Bruno Christ, Uniklinikum Leipzig
- Jürgen Reichenbach, Uniklinikum Jena
- Karl-Heinz Herrmann, Uniklinikum Jena
Contact

Tim Ricken
Univ.-Prof. Dr.-Ing.Head of Department